LGR5: A Potential Target for Next-Generation Cancer Therapies
Recently, Trends in Cancer published a review on the role of LGR5 in tumor biology. The study, conducted by researchers from UTHealth Houston and MD Anderson Cancer Center, highlights how LGR5 drives tumor plasticity and therapeutic resistance, and further explores emerging strategies for targeting LGR5. The review indicates that LGR5 is not only a key marker of cancer stem cells, but also a critical regulatory node underlying tumor plasticity and treatment failure.
Overview of LGR5
LGR5 is a Class A rhodopsin-like G protein-coupled receptor (GPCR). It is widely recognized as a stem cell marker and has increasingly important roles in cancer progression and therapeutic resistance.
LGR5 was initially identified as an orphan receptor. Structurally, it consists of seven transmembrane domains, a large extracellular domain (ECD) containing 17 leucine-rich repeats, and an intracellular C-terminal tail.
Under normal physiological conditions, LGR5 serves as a marker of crypt base columnar (CBC) cells distributed along the gastrointestinal tract. These intestinal stem cells (ISCs) exhibit high proliferative capacity and can differentiate into various cell types within the intestinal villi. Further studies have shown that LGR5 also marks a distinct population of tumor cells—cancer stem-like cells (CSCs). CSCs represent a small subset of cells within tumors with unlimited self-renewal and proliferative potential, capable of generating diverse cell types in tumors, and are considered a key driver of tumor initiation, progression, and recurrence.
Signaling Regulation Mechanism of LGR5
Amplification of Wnt/β-catenin signaling
The core function of LGR5 is to enhance Wnt/β-catenin signaling by binding to R-spondin ligands. This pathway is involved in tumorigenesis and plays a key role in regulating physiological processes, including embryonic development, stemness, and tissue homeostasis.
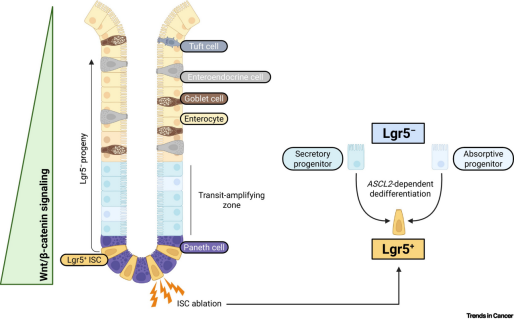
Unlike classical GPCRs, LGR5 does not transduce signaling through G proteins or β-arrestin. Although its interaction with RNF43/ZNRF3 remains debated, studies suggest that LGR5 can directly associate with the Wnt receptor complex (FZD/LRP). This interaction is markedly enhanced in the presence of both RSPO and Wnt ligands, leading to strong activation of downstream signaling.
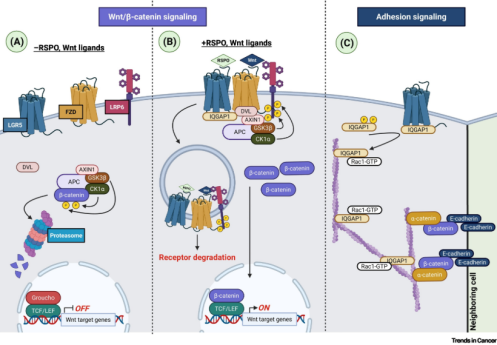
Regulation of Cell–Cell Adhesion
In addition to Wnt signaling, LGR5 also regulates cell–cell adhesion, which is important for the adaptability of stem cells and contributes to cancer cell plasticity.
LGR5-positive CSCs exhibit phenotypic plasticity, meaning they can reversibly transition between different cellular states. This plasticity enables cancer cells to respond to chemotherapy or targeted therapies (such as EGFR/KRAS/MAPK inhibitors) by switching between LGR5-positive and LGR5-negative states, thereby contributing to therapeutic resistance. Through modulation of adhesion signaling, LGR5 plays a key role in maintaining stem cell homeostasis and facilitating tumor cell adaptation to environmental changes.
Constitutive Endocytosis: A Key Mechanism for Functional Regulation and Therapeutic Targeting
A distinctive feature of LGR5 is its continuous and rapid constitutive endocytosis followed by lysosomal degradation. This process depends on clathrin and dynamin and occurs independently of RSPOs and β-arrestin. Studies have shown that deletion of the C-terminal tail inhibits LGR5 endocytosis and recycling, while increasing both basal and RSPO1-induced Wnt/β-catenin activity.
These findings suggest that endocytosis may not be required for LGR5-mediated pathway potentiation, but instead functions as a negative feedback mechanism that fine-tunes signaling activity. In addition, this highly efficient internalization property makes LGR5 an attractive target for antibody-drug conjugates (ADCs), as therapeutic agents can be efficiently delivered into cells via receptor-mediated endocytosis, providing a promising strategy for targeted cancer therapy.
Therapeutic Strategies Targeting LGR5
Current therapeutic approaches targeting LGR5 include antibody-drug conjugates (ADCs), CAR-T cell therapies, bispecific antibodies, and combination therapies. The emergence of these innovative strategies highlights the broad potential of LGR5 in cancer treatment and further underscores its central role as a key therapeutic target across multiple malignancies.
Last September, Genmab completed its acquisition of Merus for a total value of $8 billion. A key asset in the deal is petosemtamab, an EGFR/LGR5 bispecific antibody currently in Phase III clinical development. This drug, which has received Breakthrough Therapy Designation from the FDA, is becoming a central component of Genmab’s late-stage pipeline.
Petosemtamab is an EGFR×LGR5 bispecific antibody with enhanced ADCC activity and has the potential to become a “first-in-class and best-in-class” therapy for head and neck cancers. Mechanistically, upon binding to tumor cells, EGFR is targeted for degradation via the LGR5/E3 ubiquitin ligase pathway. In addition, the antibody can recognize and bind LGR5-positive stem-like cells without interfering with R-spondin ligand binding or signaling, thereby minimizing disruption to normal LGR5-positive stem cells.
At ASCO 2025, Phase II data in head and neck cancer showed an objective response rate (ORR) of 60% and a 6-month overall survival (OS) rate of 93% when combined with PD-1 therapy. The drug has received two FDA Breakthrough Therapy Designations and is currently in Phase III trials, with the potential to become a new standard of care in head and neck cancer.
Two parallel Phase III clinical trials are underway for first-line and second/third-line treatment of head and neck squamous cell carcinoma (HNSCC), with interim top-line data from one or both studies expected this year.
Genmab has indicated that, if clinical outcomes meet expectations and regulatory approval is obtained, the drug could achieve its first market launch in 2027. The company also plans to further expand the development scope of petosemtamab and accelerate its clinical program, including exploration of earlier-line treatment settings.
Outstanding Challenges and Future Directions in Targeting LGR5
However, despite these advances, important challenges remain in fully translating LGR5 biology into durable clinical benefit. As highlighted in the review, while LGR5 clearly modulates Wnt/β-catenin signaling and cell adhesion programs, the precise molecular mechanisms underlying its signaling potentiation are still not fully resolved, and key components of its functional network remain under active investigation. Moreover, although LGR5-targeted therapies—including ADCs, CAR-T approaches, and bispecific antibodies—have demonstrated encouraging antitumor activity across multiple tumor types, tumor recurrence after treatment continues to be observed, underscoring the persistence of therapy-resistant cellular populations and intratumoral heterogeneity.
Emerging evidence further suggests that therapeutic pressure can dynamically reshape LGR5 expression states, contributing to reversible transitions between LGR5-positive and LGR5-negative tumor cell populations. This plasticity may represent a central mechanism of adaptive resistance, particularly in the context of chemotherapy and targeted agents such as EGFR, KRAS, and MAPK inhibitors. These findings support the rationale for combination or multitarget strategies, including dual targeting of LGR5 with pathways such as EGFR, to more effectively address tumor heterogeneity and prevent relapse.
Taken together, while accumulating preclinical and clinical data strongly support LGR5 as a high-value therapeutic target, its context-dependent biology and role in tumor cell plasticity highlight the need for deeper mechanistic understanding and more refined therapeutic designs to achieve durable tumor eradication.
About Genomeditech
To address the key challenges highlighted in recent LGR5 research, Genomeditech offers a comprehensive portfolio of LGR5-expressing cell lines designed specifically for target mechanism studies and drug development.
Engineered for stable and consistent LGR5 expression, these cell models provide a dependable platform for investigating the R-spondin–LGR5 axis, Wnt/β-catenin pathway activation, and downstream signaling dynamics. They also support applications such as receptor internalization analysis, membrane trafficking studies, and protein interaction characterization—enabling deeper insights into LGR5 biology.
In drug discovery, these cell lines facilitate efficient evaluation of chemotherapeutics, antibody-drug conjugates (ADCs), and targeted inhibitors, including EGFR, KRAS, and MAPK pathways. By enabling precise assessment of LGR5-related signaling responses and drug sensitivity, they help streamline early-stage screening and mechanism-of-action validation.
With high reproducibility, scalability, and experimental flexibility, Genomeditech’s LGR5 cell line solutions provide a standardized in vitro platform to support both fundamental research and translational applications—accelerating the development of next-generation LGR5-targeted therapies.
High, P. C., Cappellino, M. G., Blackburn, T. A., & Carmon, K. S. (2026). LGR5: from stem cell marker to therapeutic target. Trends in Cancer. https://doi.org/10.1016/j.trecan.2026.02.006
Genmab to Acquire Merus, Expanding Late-Stage Pipeline and Accelerating into a Wholly Owned Model. (2025). Genmab. https://ir.genmab.com/news-releases/news-release-details/genmab-acquire-merus-expanding-late-stage-pipeline-and/
Merus’ Petosemtamab with Pembrolizumab Interim Data Demonstrates Robust Efficacy and Durability in 1L PD-L1+ r/m HNSCC - Merus. (2025). Merus. https://ir.merus.nl/news-releases/news-release-details/merus-petosemtamab-pembrolizumab-interim-data-demonstrates