On April 7, 2026, Sanofi announced that its investigational bispecific Nanobody® therapy lunsekimig achieved primary and key secondary endpoints in Phase 2 studies for moderate-to-severe asthma and chronic rhinosinusitis with nasal polyps (CRSwNP), while demonstrating a favorable safety profile across trials. Designed to simultaneously inhibit thymic stromal lymphopoietin (TSLP), an upstream initiator of type 2 inflammation, and IL-13, a key downstream effector driving tissue damage, lunsekimig showed significant reductions in asthma exacerbations and improvements in lung function, as well as meaningful reductions in nasal polyp burden and symptom scores. These results highlight the therapeutic potential of targeting TSLP as a central regulator of inflammatory pathways in respiratory diseases.
Overview of the TSLP Target
The TSLP/TSLPR signaling pathway plays a critical role in regulating immune responses, particularly in allergic and autoimmune diseases. It is also involved in modulating T cell polarization, dendritic cell activation, and antigen presentation, highlighting its broad impact on immune cell function.
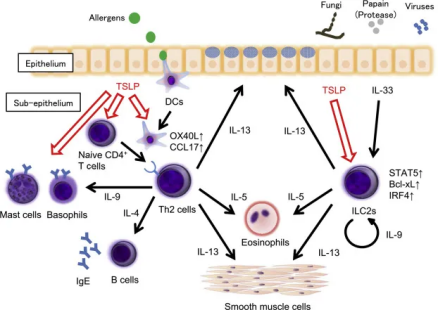
Thymic stromal lymphopoietin (TSLP) is an epithelial cell-derived cytokine that is key in the initiation and persistence of airway inflammation. TSLP drives the release of downstream type 2 (T2) cytokines, including IL-4, IL-5, and IL-13, contributing to asthma and other inflammatory symptoms. In addition, TSLP can activate multiple cell types involved in non-T2-driven inflammation.
Two isoforms of TSLP exist in human tissues. The short form (sfTSLP), consisting of 63 amino acids, is predominantly expressed under steady-state conditions and is associated with TSLP’s homeostatic functions. The long form (lfTSLP), comprising 159 amino acids, can be upregulated by Toll-like receptor ligands, pro-inflammatory cytokines, specific cellular environments, or TNF-α. Expression of the long isoform occurs upstream in multiple inflammatory cascades, mediating exaggerated immune responses in various allergic diseases, and has been shown to play a central role in type 2 immunity.
TSLP signals through its receptor, TSLPR, a member of the hematopoietic cytokine receptor family and a type I cytokine receptor. TSLPR binds TSLP with low affinity on its own but forms a high-affinity receptor complex when associated with the IL-7 receptor α chain (IL-7Rα).
Preclinical studies have shown that glucocorticoids reduce TSLP and TSLPR expression in asthma models, altering TSLP-induced dendritic cell function. This shift changes antigen-driven Th2 responses toward Th1 responses, reducing IL-4, IL-5, and IL-13 while increasing IL-10 and IFN-γ. These effects are mediated by downregulation of OX40L and increased IL-12 production in dendritic cells. Clinical data in asthma patients further indicate a positive correlation between TSLP expression levels and the severity of airway inflammation.
Overall, the TSLP-TSLPR axis and its downstream signaling molecules play a critical role in the development and progression of allergic inflammatory diseases, such as allergic asthma.
TSLP Continues to Gain Attention
TSLP is being increasingly recognized as a key target across the autoimmune and inflammatory disease landscape. Evidence has linked TSLP to a range of allergic diseases, including atopic dermatitis, bronchial asthma, and eosinophilic esophagitis. Emerging studies also suggest its involvement in chronic inflammatory conditions such as chronic obstructive pulmonary disease (COPD) and celiac disease, as well as autoimmune disorders including psoriasis and rheumatoid arthritis.
Currently, the only approved TSLP-targeting monoclonal antibody on the market is tezepelumab. Since its launch, the drug has rapidly scaled, becoming a blockbuster with annual sales exceeding one billion USD. In 2024, sales reached approximately $1.22 billion, followed by around $936 million in 2025, directly validating the commercial potential of the TSLP target and expanding the market’s outlook for the entire therapeutic area. Market analyst Leerink forecasts that tezepelumab could achieve sales of roughly $4.3 billion by 2030.
From an R&D perspective, the TSLP target is in a phase of rapid expansion. Efforts span from traditional monoclonal antibodies to bispecifics, trispecifics, long-acting formulations, and even inhaled delivery, reflecting a nearly comprehensive exploration of TSLP-targeted strategies.
In terms of multi-antibody development, TSLP is being combined with IL-4, IL-13, IL-4Rα, and related pathways. Notable programs include Innovent’s IBI3002 (IL-4Rα/TSLP), Sanofi’s lunsekimig (IL-13/TSLP), CanNovia’s CM512 (TSLP/IL-13), and Pfizer’s PF-07275315, a trispecific targeting IL-4/IL-13/TSLP. Additionally, PF-07275315 is undergoing clinical trials domestically for moderate-to-severe atopic dermatitis and adult moderate-to-severe asthma.
Regarding administration routes, the standard remains every 2–4 weeks, but there is a clear trend toward long-acting formulations. Hengrui Medicine’s SHR-1905 is exploring ultra-long dosing intervals of up to six months; Kelun-Botai/Harbour BioMed’s HBM9378/SKB378 extends half-life by 2–3 times; and Amgen/AstraZeneca’s AZD8630 uses daily inhaled administration to improve patient adherence.
Conclusion
With its ability to act as an upstream regulator and broadly suppress inflammatory signaling at the source, TSLP is rapidly emerging as a key battleground for both multinational pharmaceutical companies and Chinese biotech firms. From monoclonal antibodies to bispecifics, and from long-acting formulations to inhaled therapies, a wide range of development strategies is advancing in parallel. As innovation accelerates, the therapeutic and commercial potential of this target continues to expand, positioning TSLP as a leading contender in the next wave of high-value autoimmune and inflammatory disease therapies.
Reference
ClinicalTrials.eu. (2023). Solrikitug – Application in Therapy and Current Clinical Research. https://clinicaltrials.eu/inn/solrikitug/
GSK. (2024, January 9). GSK enters agreement to acquire Aiolos Bio. https://www.gsk.com/en-gb/media/press-releases/gsk-enters-agreement-to-acquire-aiolos-bio
Sanofi. (2025, April 15). Sanofi’s respiratory pipeline advances with new data in asthma and plans for new clinical studies in COPD. https://www.sanofi.com/en/media-room/press-releases/2025/2025-04-15-05-00-00-3061368
U.S. Food and Drug Administration. (2021, December 17). FDA approves maintenance treatment for severe asthma. https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-maintenance-treatment-severe-asthma
Harbour BioMed. (2025, July 23). HBM9378/WIN378, a long-acting, fully human anti-TSLP antibody, enters global Phase 2 POLARIS trial for asthma. https://www.harbourbiomed.com/news/244.html
AstraZeneca. (n.a.). A Phase 2b, randomised, double-blind, placebo-controlled dose range-finding study to assess efficacy and safety of multiple dose levels of inhaled AZD8630 given once daily for 12 weeks in adults with uncontrolled asthma at risk of exacerbations (LEVANTE). https://www.astrazenecaclinicaltrials.com/study/D6830C00003/
ClinicalTrials.gov. (2026, January). A study to evaluate solrikitug in participants with chronic obstructive pulmonary disease (COPD) (ZION). U.S. National Library of Medicine. https://clinicaltrials.gov/study/NCT06496620
ClinicalTrials.gov. (2025, May). A study to learn about the study medicine called PF-07275315 in people with moderate-to-severe asthma. U.S. National Library of Medicine. https://clinicaltrials.gov/study/NCT06977581