Recent developments have brought renewed attention to the IL-17 pathway in inflammatory disease research. Clinical data released by UCB indicate that dual IL-17A/F inhibition may offer improved efficacy compared to single-target approaches from AbbVie, while Novartis has expanded the clinical reach of Secukinumab into new patient populations.
Taken together, these advances suggest that even well-established immunology targets such as IL-17 continue to evolve, both mechanistically and clinically.
IL-17 Signaling Pathway: Key Nodes Bridging Upstream and Downstream
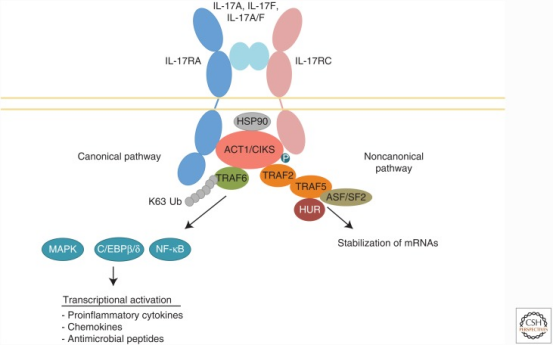
Interleukin-17A (IL-17A) is the prototypical member of the IL-17 cytokine family and is primarily produced by Th17 cells. It plays a central role in orchestrating inflammatory responses, not by initiating them, but by amplifying downstream signaling cascades.
A defining feature of this pathway is its structural and functional redundancy. IL-17A shares high homology with IL-17F, and the two cytokines can form homodimers as well as IL-17A/F heterodimers. These ligands signal through receptor complexes composed of IL-17RA and IL-17RC, activating adaptor proteins such as ACT1 and triggering canonical pathways including NF-κB and MAPK.
This signaling cascade ultimately induces the production of pro-inflammatory mediators such as IL-6, IL-8, and GM-CSF across multiple cell types, including epithelial cells and fibroblasts. From a translational perspective, IL-17 occupies a unique position: it is sufficiently downstream to ensure a direct effect on inflammation, yet central enough to produce broad biological impact when inhibited.
From Single to Dual Targeting: Mechanism-Driven Evolution
The first generation of IL-17-targeted therapies primarily focused on IL-17A alone, achieving strong clinical outcomes in diseases such as psoriasis and psoriatic arthritis. However, deeper mechanistic understanding has revealed that IL-17F remains biologically active even when IL-17A is neutralized, allowing residual signaling to persist.
This insight has driven the development of dual IL-17A/F inhibitors, which aim to more completely suppress pathway activity by targeting all relevant ligand combinations. The rapid clinical and commercial emergence of Bimekizumab illustrates how addressing cytokine redundancy can translate into differentiated efficacy.
Rather than representing a shift to a new target, this evolution reflects a refinement of an already validated pathway, where incremental mechanistic gains may yield meaningful clinical benefits.
A Mature Yet Expanding Market
The IL-17 class has transitioned from innovation to establishment, yet it continues to expand across both indications and geographies. Therapies targeting IL-17A have set a strong foundation in chronic inflammatory diseases, and lifecycle strategies are now extending their relevance into additional conditions such as hidradenitis suppurativa and axial spondyloarthritis.
At the same time, next-generation molecules are redefining competition within the class. Improvements in efficacy, dosing convenience, and durability are becoming key differentiators, while mechanistic innovation remains focused on deeper and more sustained pathway suppression.
This combination of clinical validation and ongoing optimization has positioned IL-17 as one of the most resilient and commercially durable targets in immunology.
Global R&D Landscape: Convergence and Differentiation
Across regions, the IL-17 pipeline reflects both convergence on validated biology and divergence in development strategies. While many programs continue to pursue IL-17A inhibition, an increasing number are exploring dual-target approaches, antibody engineering, and extended dosing regimens.
In emerging biopharmaceutical markets, R&D activity in this field is also continuing to evolve and intensify.

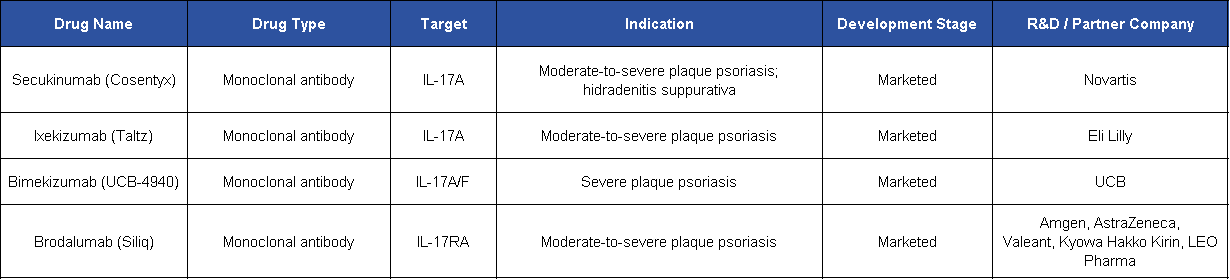
Biocad’s IL-17A monoclonal antibody, Netakimab, has already been approved in Russia. Meanwhile, Livzon Pharmaceutical’s LZM012 adopts a dual IL-17A/F targeting strategy, similar to UCB’s bimekizumab, offering potential mechanistic advantages over single-target therapies. Notably, it is the first IL-17A/F dual inhibitor in China—and the second globally—to reach Phase III clinical development.
At the same time, increasing competition driven by patent expirations is reshaping the market landscape, where originator biologics, next-generation improved therapies, and biosimilars are expected to coexist.
In this evolving environment, R&D priorities are shifting—from target selection toward deeper mechanistic understanding and improved translational efficiency.
Translational Considerations in IL-17 Drug Development
Despite its maturity, the IL-17 pathway continues to present challenges at the translational level. The redundancy of cytokine networks complicates target validation, while the complexity of inflammatory microenvironments makes it difficult to fully recapitulate disease biology in preclinical models.
Bridging the gap between in vitro systems and clinical outcomes remains a critical step. Functional assays that capture pathway activation, receptor engagement, and downstream signaling are essential for de-risking development. In this context, robust cellular models and pathway-relevant systems play an increasingly important role in improving predictability and efficiency.
Reference
Novartis. (2026, March 13). Novartis Cosentyx receives FDA approval for pediatric patients aged 12+ with moderate to severe hidradenitis suppurativa. https://www.novartis.com/news/media-releases/novartis-cosentyx-receives-fda-approval-pediatric-patients-aged-12-moderate-severe-hidradenitis-suppurativa
Novartis. (2023, October 31). FDA approves Novartis Cosentyx as the first new biologic treatment option for hidradenitis suppurativa patients in nearly a decade. https://www.novartis.com/news/media-releases/fda-approves-novartis-cosentyx-first-new-biologic-treatment-option-hidradenitis-suppurativa-patients-nearly-decade
Eli Lilly and Company. (2020, March 30). Lilly’s Taltz (ixekizumab) receives U.S. FDA approval for treatment. https://investor.lilly.com/news-releases/news-release-details/lillys-taltzr-ixekizumab-receives-us-fda-approval-treatment-1
UCB. (2024, November 20). UCB receives U.S. FDA approval for Bimzelx (bimekizumab-bkzx) as the first IL-17A and IL-17F inhibitor for adults with moderate-to-severe hidradenitis suppurativa. https://www.ucb-usa.com/stories-media/UCB-U-S-News/detail/article/ucb-receives-us-fda-approval-for-bimzelxr-bimekizumab-bkzx-as-the-first-il-17a-and-il-17f-inhibitor-for-adults-with-moderate-to-severe-hidradenitis-suppurativa
AstraZeneca. (2017, February 16). Siliq (brodalumab) approved by the U.S. FDA for adult patients with moderate-to-severe plaque psoriasis. https://www.astrazeneca.com/media-centre/press-releases/2017/Siliq-brodalumab-approved-by-the-US-FDA-for-adult-patients-with-moderate-to-severe-plaque-psoriasis-160220170.html
FirstWord Pharma. (2019, May 09). BIOCAD registered the first Russian original therapeutic monoclonal antibody. https://firstwordpharma.com/story/4787481
ClinicalTrials.gov. (2023, November 01). A Study to Evaluate the Efficacy and Safety of LZM012 (Psoriasis). https://clinicaltrials.gov/study/NCT06110676
Monin L, & Gaffen SL (2018). Interleukin 17 family cytokines: Signaling mechanisms, biological activities, and therapeutic implications. Cold Spring Harbor Perspectives in Biology, 10(4), a028522. https://doi.org/10.1101/cshperspect.a028522