Interleukin-23 (IL-23) is an important pro-inflammatory cytokine belonging to the interleukin-12 family. It is produced by antigen-presenting cells such as macrophages and dendritic cells.
IL-23 is composed of two subunits: IL-12p40 and IL-23p19. The p40 subunit is shared with IL-12, while p19 is the function-specific subunit unique to IL-23. By binding to the IL-23R/IL-12Rβ1 receptor complex, IL-23 activates intracellular signaling pathways and promotes the proliferation and differentiation of Th17 cells, a process that plays a central role in the initiation and amplification of inflammatory responses.
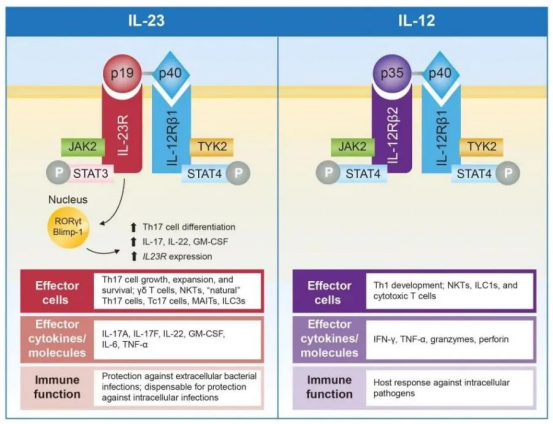
The IL-23 signaling pathway involves two receptor chains and intracellular signaling proteins, including Jak2 and Tyk2, as well as the downstream JAK targets STAT3 and STAT4. Upon stimulation by IL-23, the receptor complex activates Jak2 and Tyk2, leading to phosphorylation of the receptor and the formation of docking sites for STAT proteins. STAT proteins are subsequently phosphorylated, dimerized, and translocated into the nucleus, where they activate target gene transcription. Phosphorylation of STAT4 is critical for IFN-γ secretion and subsequent Th1 cell differentiation, while phosphorylation of STAT3 is essential for the development of Th17 cells.
Overall, activation of the IL-23 pathway induces cytokine cascades through STAT3 and STAT4 signaling, enhances immune cell activity, and promotes the persistence of inflammatory responses.
A Key Driver of Autoimmune Disease
IL-23 is a major cytokine that links innate and adaptive immune responses. It promotes early mucosal defense and amplifies local inflammation, playing a critical regulatory role during the initial phase of immune activation.
Early studies also demonstrated that IL-23 can induce IFN-γ production, a cytokine essential for Th1 responses and cell-mediated immunity against intracellular pathogens. In addition, IL-23 plays a central role in activating NK cells, enhancing T-cell proliferation, and regulating antibody production.
Extensive clinical and preclinical evidence indicates that elevated IL-23 levels are associated with multiple autoimmune diseases, including psoriasis, inflammatory bowel disease (IBD), rheumatoid arthritis (RA), and multiple sclerosis (MS).
Importantly, IL-23 is a key maturation and maintenance factor for Th17 cells, which are major effectors driving chronic autoimmune inflammation. As an upstream regulator of the IL-17 pathway, IL-23 represents a critical signaling node in autoimmune pathogenesis and has emerged as a major therapeutic target in the development of treatments for autoimmune diseases.
Global Landscape of IL-23 Therapeutics and Emerging Pipelines
With the rapid advancement of therapies for autoimmune diseases, IL-23 has become a key target attracting extensive investment and strategic positioning by pharmaceutical companies worldwide.
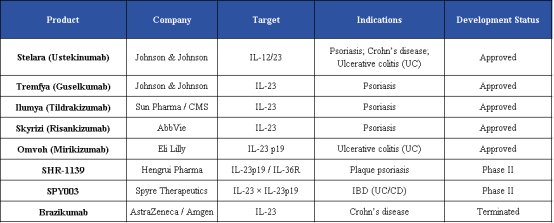
Globally, multiple IL-23–targeting antibody drugs have been approved and can be broadly divided into two categories:
1) IL-23 p19 subunit inhibitors
Five IL-23 p19–targeting therapies have been approved worldwide. These include products developed by Innovent Biologics, AbbVie, CMS Pharmaceuticals, Johnson & Johnson, and Eli Lilly and Company. These agents selectively block the IL-23 p19 subunit, enabling targeted inhibition of IL-23 signaling.
2) IL-12/23 p40 dual inhibitors
A second class targets the shared p40 subunit of IL-12 and IL-23, thereby inhibiting IL-23 signaling while also modulating IL-12–mediated immune responses. Representative therapies include those developed by Akeso Biopharma and Johnson & Johnson.
A notable recent development is an IL-23 p19 antibody from Eli Lilly and Company, which received approval from the U.S. Food and Drug Administration in January 2025 for the treatment of Crohn’s disease, expanding its indications to a second major category of inflammatory bowel disease.
In the Phase III VIVID-1 trial for Crohn’s disease, the treatment group demonstrated higher rates of clinical response at Week 12 and clinical remission at Week 52 compared with placebo (45.4% vs 19.6%). Endoscopic response at Week 52 reached 38%, compared with 9% in the placebo group. While non-inferior to ustekinumab in achieving clinical remission, superiority in endoscopic response was not observed.
In ulcerative colitis, the Phase III LUCENT program showed similarly positive outcomes. In LUCENT-1, clinical remission at Week 12 was significantly higher than placebo (24.2% vs 13.3%, P<0.001). In the LUCENT-2 maintenance phase, remission at Week 40 was also markedly improved (49.9% vs 25.1%, P<0.001). Overall, IL-23 p19–targeted therapy has demonstrated significant efficacy in moderate-to-severe Crohn’s disease and ulcerative colitis, further validating IL-23 as a major therapeutic axis.
Beyond monoclonal antibodies, innovative pipeline strategies are emerging. A notable example is SHR-1139 developed by Jiangsu Hengrui Pharmaceuticals. As the first IL-23 dual-target therapy (IL1RL2 and IL23A) to enter Phase II clinical trials globally, this candidate may achieve broader pathway inhibition and potentially address limitations associated with single-target therapies, including resistance and suboptimal clinical response. The program entered Phase II trials on March 13, 2025, initially targeting plaque psoriasis, with additional autoimmune indications under clinical investigation.
Overall, a substantial number of IL-23–related programs remain in clinical development, highlighting the strong innovation momentum and long-term therapeutic potential of this pathway.
References
AbbVie. (2022, June 17). SKYRIZI® (risankizumab-rzaa) receives FDA approval as the first and only specific interleukin-23 (IL-23) to treat moderately to severely active Crohn’s disease in adults. https://news.abbvie.com/2022-06-17-SKYRIZI-R-risankizumab-rzaa-Receives-FDA-Approval-as-the-First-and-Only-Specific-Interleukin-23-IL-23-to-Treat-Moderately-to-Severely-Active-Crohns-Disease-in-Adults
ClinicalTrials.gov. (n.d.). Study of SPY003 in participants with ulcerative colitis (NCT07012395). https://clinicaltrials.gov/study/NCT07012395
ClinicalTrials.gov. (n.d.). A study evaluating treatment in inflammatory bowel disease (NCT07232576). https://clinicaltrials.gov/study/NCT07232576
Eli Lilly and Company. (2024). FDA approves Lilly’s Omvoh™ (mirikizumab-mrkz) for Crohn’s disease. https://investor.lilly.com/news-releases/news-release-details/fda-approves-lillys-omvohr-mirikizumab-mrkz-crohns-disease
Johnson & Johnson. (2023). Johnson & Johnson seeks U.S. FDA approval of STELARA® (ustekinumab) for the treatment of pediatric Crohn’s disease. https://www.jnj.com/media-center/press-releases/johnson-johnson-seeks-u-s-fda-approval-of-stelara-ustekinumab-for-the-treatment-of-pediatric-crohns-disease
Johnson & Johnson. (2024). TREMFYA® (guselkumab) receives U.S. FDA approval for adults with moderately to severely active ulcerative colitis. https://www.jnj.com/media-center/press-releases/tremfya-guselkumab-receives-u-s-fda-approval-for-adults-with-moderately-to-severely-active-ulcerative-colitis-strengthening-johnson-johnsons-leadership-in-inflammatory-bowel-disease
Fierce Biotech. (2024). AstraZeneca drops Skyrizi rival after seeing R&D timeline spiral. https://www.fiercebiotech.com/biotech/astrazeneca-drops-skyrizi-rival-after-seeing-rd-timeline-spiral
Sun Pharmaceutical Industries Ltd.. (2020). Sun Pharma announces U.S. FDA approval of ILUMYA™ (tildrakizumab-asmn). https://sunpharma.com/wp-content/uploads/2020/12/Sun-Pharma-Announces-U.S.-FDA-Approval-of-ILUMYA.pdf